PIPELINE
Pharmaceutical Solutions Based on
Long-acting Technology
Long-acting Technology
Pipeline
Immunoforge specializes in novel drug development with pipelines based on half-life extension platform technology.
- Home
- >
- Pipeline
- >
- Pipeline
Main Pipelines
| Identification | Indication | Target | Discovery | Non-clinical | Phase I | Phase II | Comments | |
|---|---|---|---|---|---|---|---|---|
| ELP Platform |
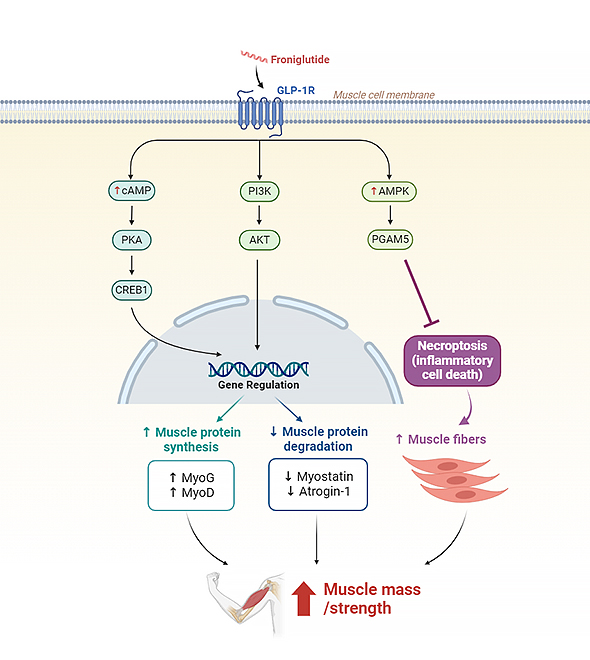
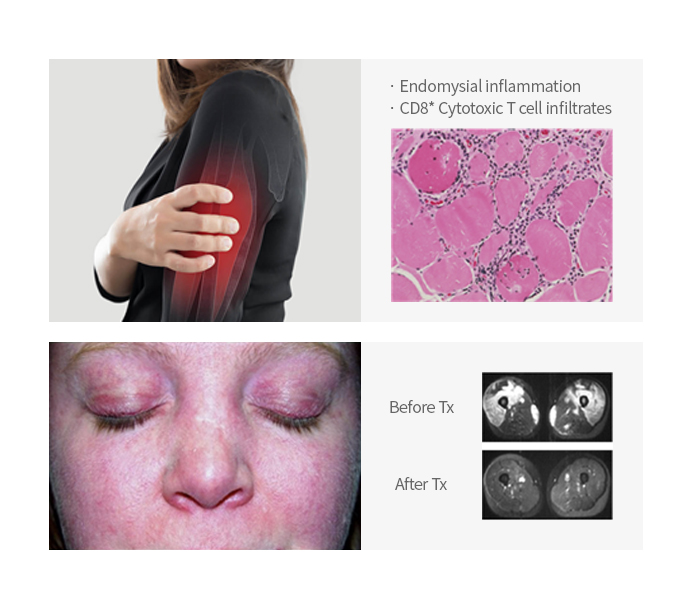
Froniglutide | Dermatomyositis (DM)/ Polymyositis (PM) |
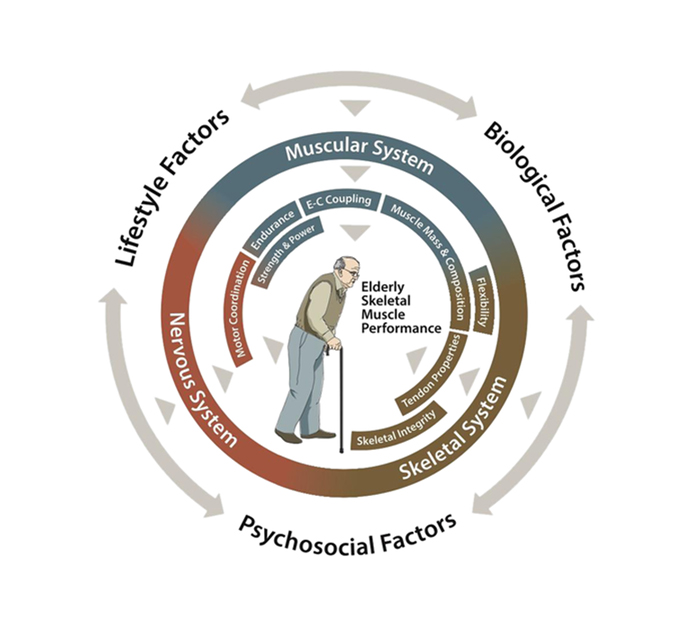
GLP-1R* (Weekly) * Sarcopenia |
|
FDA, MFDS ODD |
|||
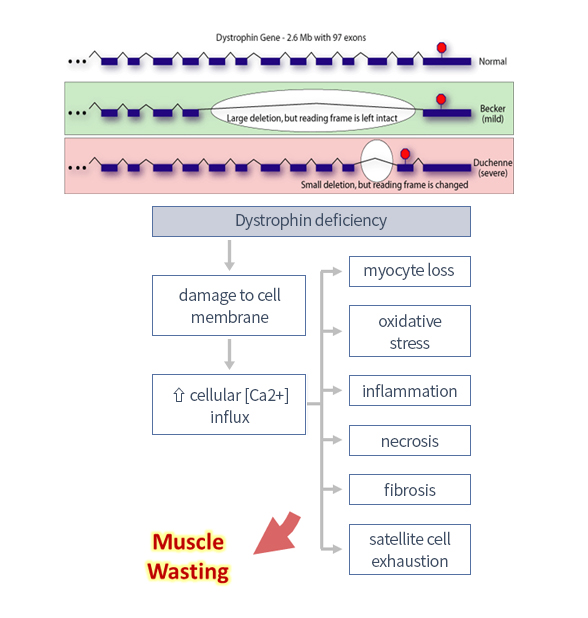
| Duchenne Muscular Dystrophy (DMD) |
|
FDA ODD | ||||||
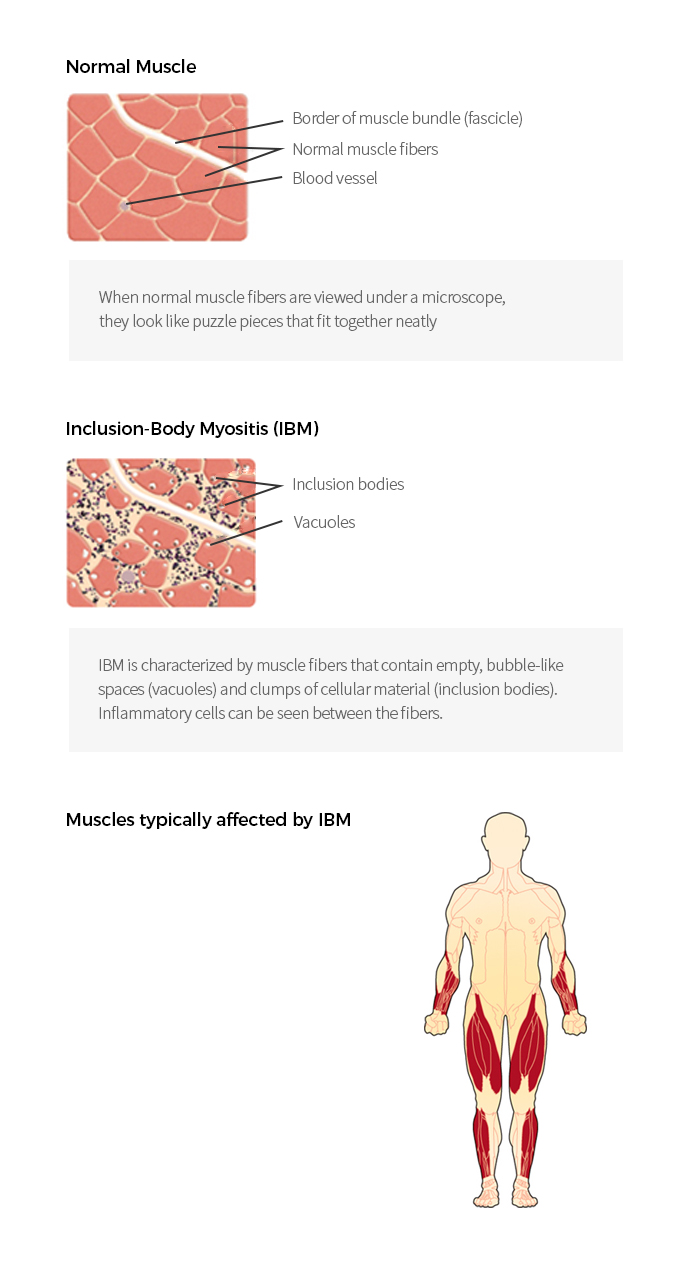
| Inclusion Body Myositis (IBM) |
|
|||||||
| Senile Sarcopenia |
|
|||||||
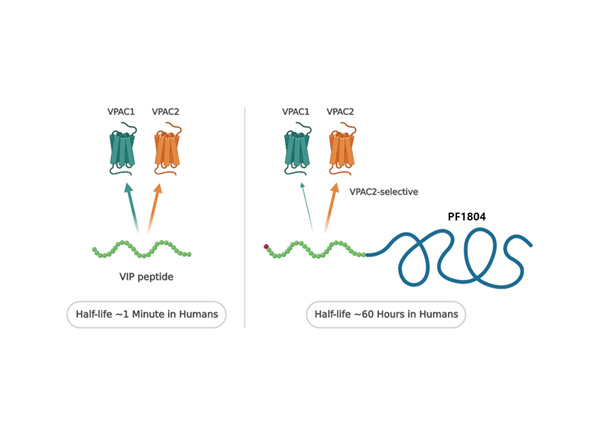
| Pemziviptadil | Cardiomyopathy(DMD), Heart Failure, Cystic Fibrosis |
VPAC2 |
|
FDA ODD | ||||
| PF1805 | Achondroplasia | NPR-B (Weekly) |
|
|||||
| PF1806 | Short Bowel Syndrome (SBS) | GLP-2R (Weekly) |
|
|||||
| PF1807 | Sarcopenia | GLP-1R (Monthly) |
|
|||||
| PF1808 | L/O |
|
||||||
| PF1802 | Amyotrophic Lateral Sclerosis (ALS), Parkinson’s Disease |
Bifunctional |
|
|||||
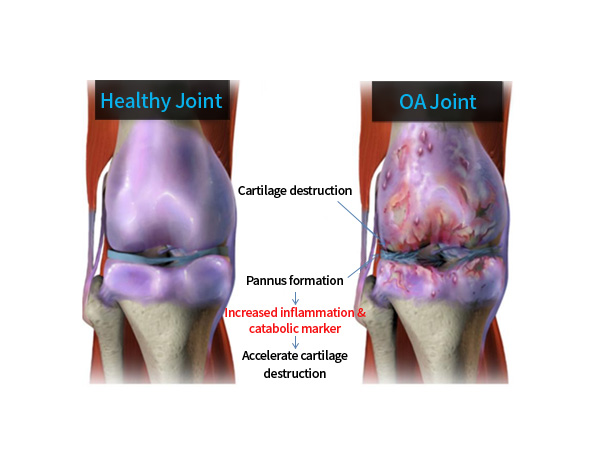
| PF1803 | Osteoarthritis (OA) | Bifunctional OSCAR + |
|
|||||
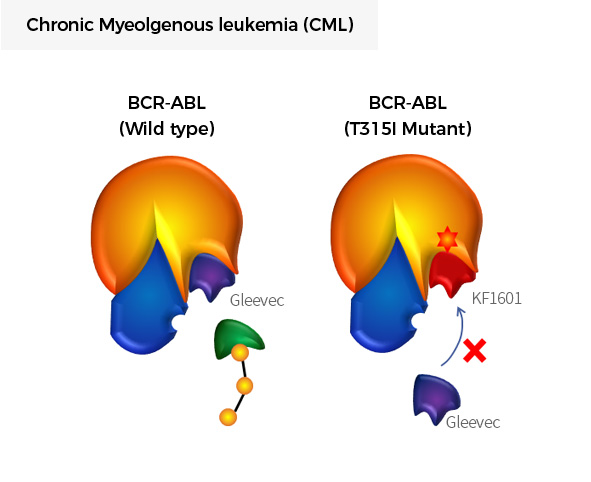
| KF1601 | Drug Resistant Chronic Myeloid Leukemia (CML) |
BCR-ABL1 (T315I) |
|
FDA ODD | ||||